You want to identify new biomarkers or determine antibody specificities?
engine Protein Arrays — Explore thousands of interactions in one step!
engine is your reliable partner for the analysis of your sample. Our protein array screening service covers the analysis of positive interactions up to antigen identification. In a single experiment it is possible to screen your sample for interactions with more than 10,000 antigens. Our fast method is non-biased and guarantees maximum outcome. You get your results much faster than with standard methods and benefit from expert support. The results and data are reliable and reproducible, will be provided to you in a confidential scientific report and can be discussed with our experts.
The core of our service are our protein arrays, which are part of more than 100 publications from different research groups. Our arrays contain more than 10,000 human proteins and peptides from various tissues and have been developed on the basis of 15 years of intensive research.
Investigate protein-antibody, protein-ligand, or protein-drug interactions, e.g., for
- highly complex autoantibody profiles
- specificity and cross-reactivity of antibodies
- protein-protein interactions for the analysis of metabolic or regulatory pathways
A wide range of samples may be used, such as
- blood sera or plasma
- cerebrospinal fluid (CSF)
- synovial fluid
- cell supernatant
Contact
More information
Stay Tuned
Discover Novel Biomarkers
Increase your chance for decisive discoveries & shorten your time to result!
Your Question. Our Arrays. Your Result!
Are you passionate to discover new biomarkers for advancing diagnostics or predicting therapy success? Until now, you usually had to use different and complex methods depending on the sample and the antigens to be tested. Those times are over! With our Protein array screening service we help you to explore similarities or differences between groups of patients, diseased or healthy individuals.
- Give us your hypothesis and sample.
- We analyze interactions with >10,000 antigens with less than 50µl sample volume in any matrix.
- You identify antigens and proceed with biomarker validation.
The unbiased, hypothesis-free analysis has a high discovery power to detect novel biomarkers and allows an objective analysis of causal disease-correlations. Compare biospecimens to identify disease-related associations or investigate if protein-modifications are linked to a disease or are causative. With ready-to-use results you can go on promptly with your biomarker validation.


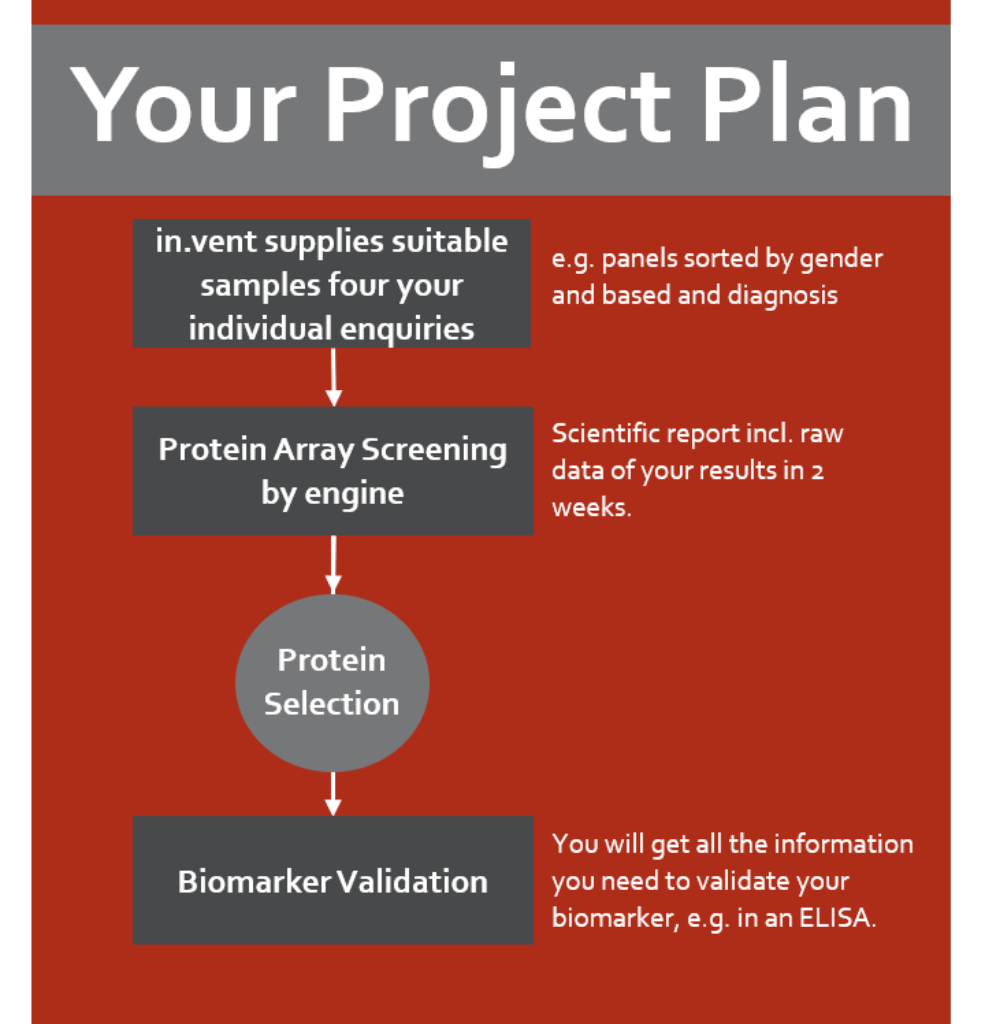
Your full-service biomarker package
Together with in.vent, we identify and validate your biomarker — from sample to upscaling.
Analyze autoantibody profiles of different patient cohorts, so you can discover disease-specific biomarkers. in.vent provides you with specific sample panels e.g. for Crohn’s disease, diabetes type I & II, rheumatoid arthritis, Sjögren’s syndrome, systemic lupus erythematosus, ulcerative colitis.
Your advantages:
- procurement and assembly of human biospecimen
- protein array screening & detailed analysis
- upscaling of sample number and differentiation
- short time and cost-effective project management and realisation
- logistics, material transfer and array screening at one site
- laboratory experiments and clinical studies can be performed in parallel